|
The former definition of the mole was that a mole was 12 g of 12C contains 1 mole of 12C atoms (its molar mass is 12 g/mol). Per the amu definition, a single 12C atom weighs 12 amu (its atomic mass is 12 amu). Converting the mass, in grams, of a substance to moles requires a conversion factor of (one mole of substance/molar mass of. If the mass of a substance is known, the number of moles in the substance can be calculated. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The molar mass of an element is found on the periodic table, and it is the element's atomic weight in grams/mole (g/mol). (credit: modification of work by Mark Ott).īecause the definitions of both the mole and the atomic mass unit are based on the same reference substance, 12C, the molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. From left to right (bottom row): 32.1 g sulfur, 28.1 g silicon, 207 g lead, and 118.7 g tin. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in Cl: Molar Mass (g/mol) Cl (Chlorine) 1 × 35.453 35.453. From left to right (top row): 65.4 g zinc, 12.0 g carbon, 24.3 g magnesium, and 63.5 g copper. 
These relative weights computed from the chemical equation are sometimes called equation weights.\) atoms -1.00 mol of atoms. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. 
If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. This site explains how to find molar mass. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. 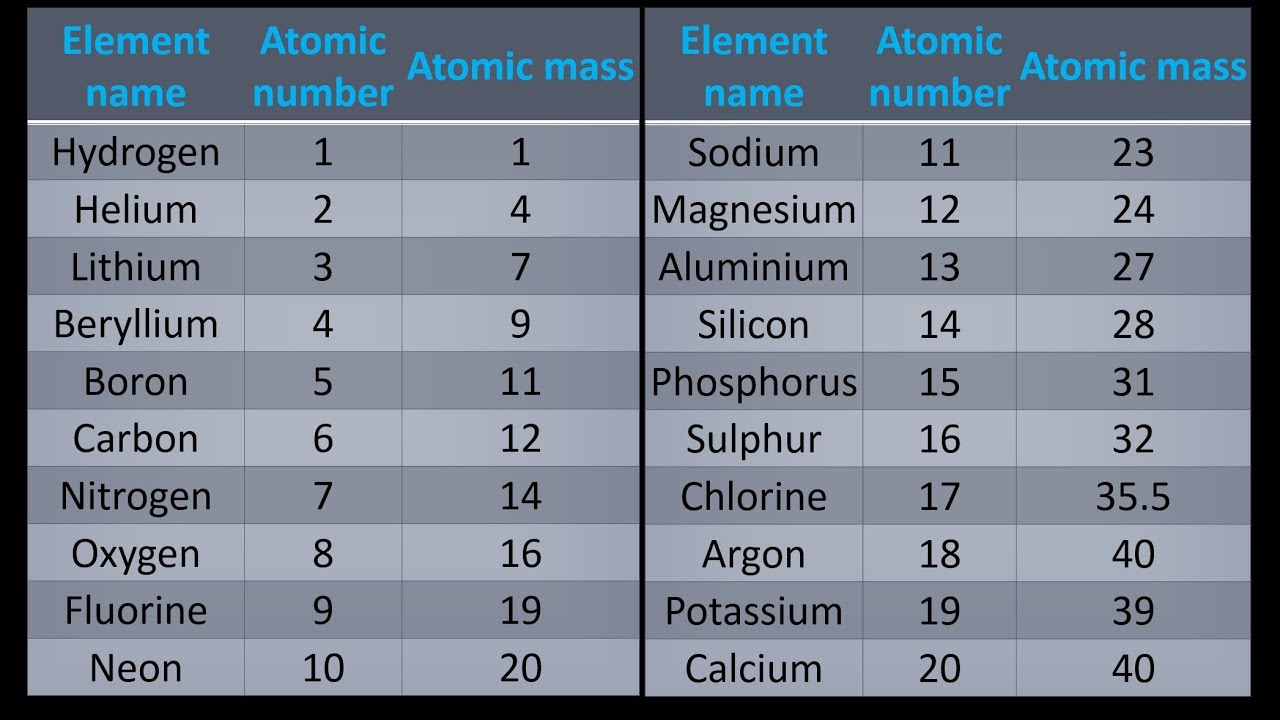
The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.Ī common request on this site is to convert grams to moles. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.įinding molar mass starts with units of grams per mole (g/mol). In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
